Temperature control in clinical trials
A clinical trial is a type of clinical trial in which results are collected under the guidance of the Ministry of Health, Labor and Welfare in order to development new drugs and obtain national approval. Clinical trials include Phase I trials (healthy adults), Phase II trials (small number of patients), Phase III trials (large number of patients), and post-marketing clinical trials (larger number of patients). When conducting clinical trials, you must follow the "Act to partially amend the Pharmaceutical Affairs Act, etc. (Pharmaceutical and Medical Device Act)" (Act No. 84 of 2013) and the "Ministry Ordinance on Standards for Conducting Clinical Trials of Pharmaceuticals (GCP Ministerial Ordinance)" (Ministry of Health, Labor and Welfare Ordinance No. 28 of March 27, 1997).
In order to ensure the accuracy of clinical trials and the safety of subjects, it is necessary to properly manage the investigational drug and clinical trial samples during the clinical trial. Investigational drugs and clinical trial samples are stored in medicine cabinets, refrigerators, freezers, incubators, low-temperature incubators, etc., and temperature control is performed using thermometers and temperature/humidity hygrometers. Temperature records must be kept, and any deviations in temperature must be checked and dealt with promptly in the event of a deviation. In addition, temperature records of stored investigational drugs and clinical trial samples, information on temperature deviations, etc. must be made available promptly upon request from the sponsor.
Regarding temperature control, the GCP does not provide clear standards for temperature check methods or recording methods, but strict management is required according to the clinical trial plan and characteristic of the investigational drug. In addition, there is an administrative notice from the Ministry of Health, Labor and Welfare titled "Basic principles regarding quality control of clinical tests in clinical trials," which requires quality control of investigational drugs and requires regular inspection of equipment.
In order to meet these demands, temperature control systems that employ electronic temperature loggers and paperless recorder to record temperatures and also notification deviations in temperature are becoming more common.
We have an extensive track record of delivering temperature control systems to the pharmaceutical and medical industries, and can meet a wide range of customer requirements, including temperature control, temperature monitoring, and thermometer calibration in clinical trials.
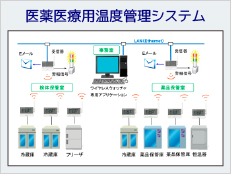
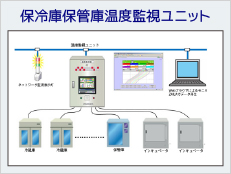
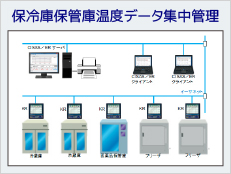
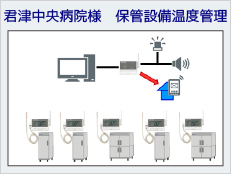
System Example
Applicable products and services
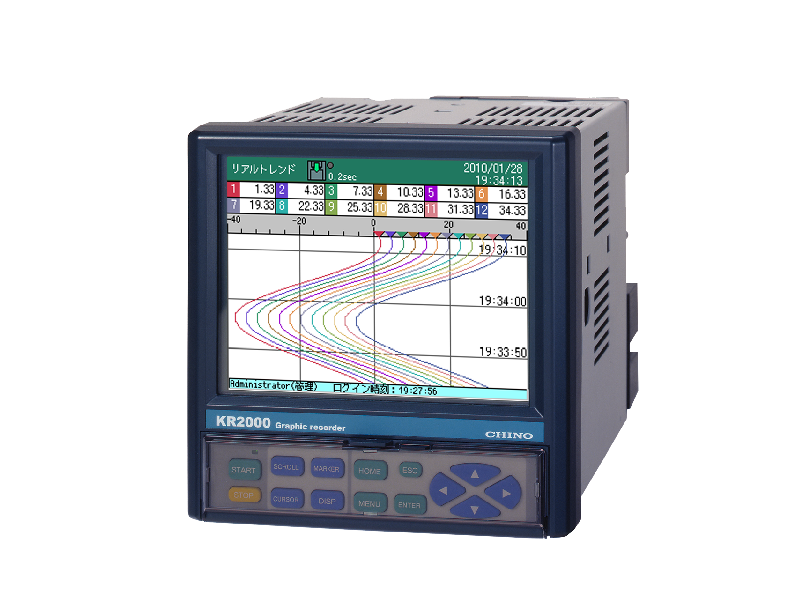
Graphic recorder with measurement data protection function KR2P00
Paperless recorder with 6 and 12 input points
Although it is small, it has a wide range display and high performance, making it suitable for use as a standalone device or small-scale monitoring equipment.
When combined with the CISAS series, it can be configured as a recorder for a computer system.
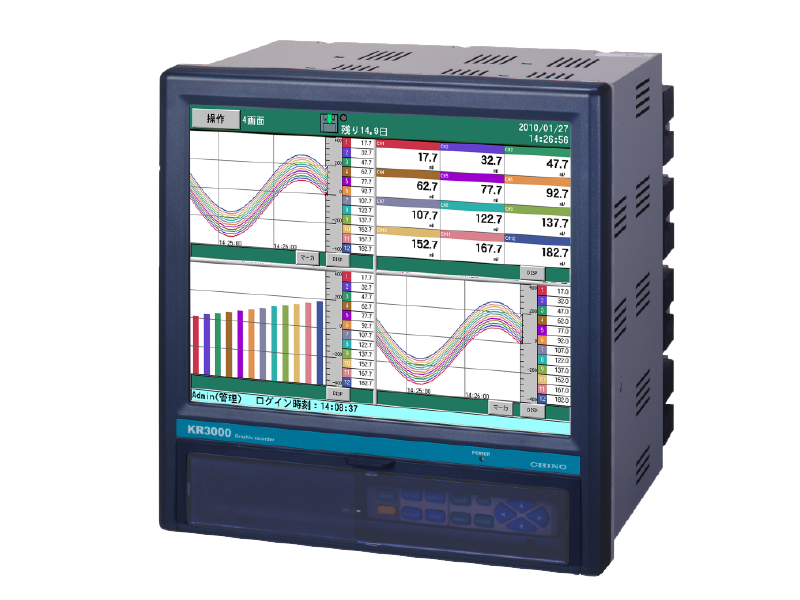
Graphic recorder with measurement data protection function KR3P00
Paperless recorder with up to 48 input points
With its versatile display and high performance, it is suitable for data management on equipment such as stability testers, refrigerators, and freezers.
When combined with the CISAS series, it can be configured as a recorder for a computer system.
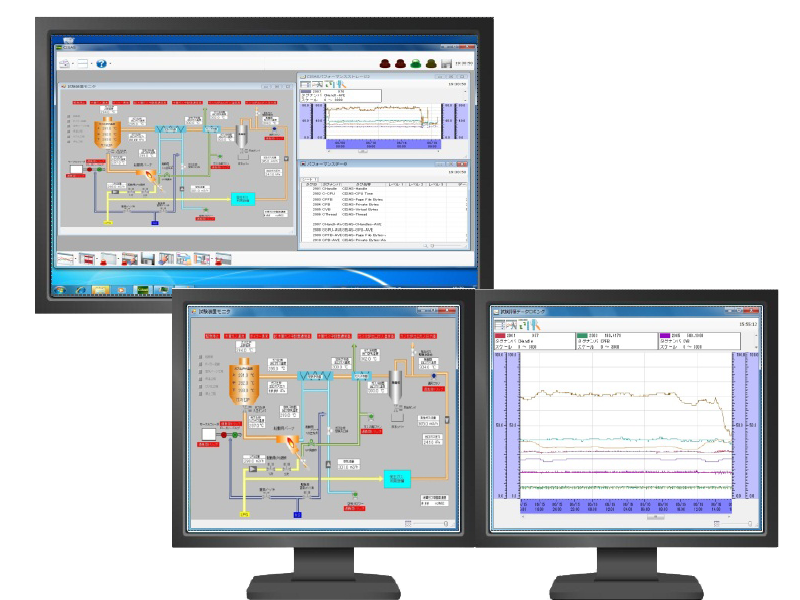
Recording and monitoring package system CISAS/V4
CISAS/V4 is a package system that uses our recorders, loggers, and controllers, as well as commercially available PLCs (programmable controllers), as system components to collect and monitor data from up to 5,000 tags of various devices and equipment on a PC.
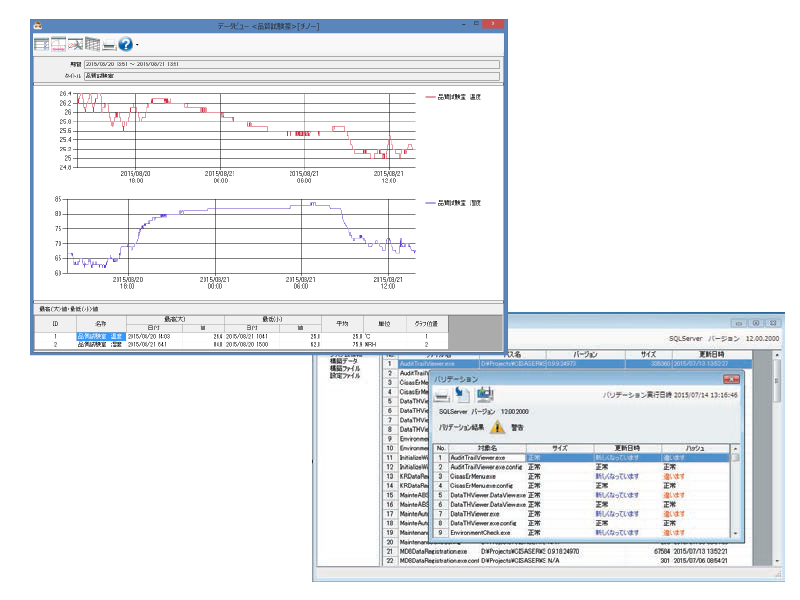
Electronic records management package software CISAS/ER
Measurement data recorded by Part 11-compliant wireless loggers and graphic recorder can be centrally managed as electronic records.
Graph display by viewing data, daily reports, monthly reports, and reports can be created (printed and output as PDF files) and electronic signatures are possible

JCSS (logo mark) calibration test
We are registration as a calibration laboratory for temperature and humidity under the Measurement Law.
We have also acquired certification as an "MRA-compliant certified business operator" based on ISO/IEC17025, and can issue calibration certificates with the JCSS certification symbol mark.

On-site inspection and calibration services
Our service technicians or contracted service personnel visit the user's site to inspect and calibrate measuring instruments, mainly temperature and humidity measuring instruments, regulators, recorders, etc.